Recommended Bills for State Implementation of Pharma Quality:
(1) Sample Evaluation of Pharmaceuticals Used in the state and (2) Random Evaluation of Pharmaceutical Manufacturing Practices of Pharmas marketing or distributing Rx Drugs
The following outline is being submitted to create legislation in South Carolina. Please contribute your ideas and comments for improvement… or not … in the comments section.
Recommended Bills for State Implementation of Pharma Quality: (1) Sample Evaluation of Pharmaceuticals Used in South Carolina and (2) Random Evaluation of Pharmaceutical Manufacturing Practices
With Appropriate, Strong and Timely Removal from Market for Either Contaminated or Mislabeled Content of Pharmaceuticals or Lack of Good Manufacturing Practices
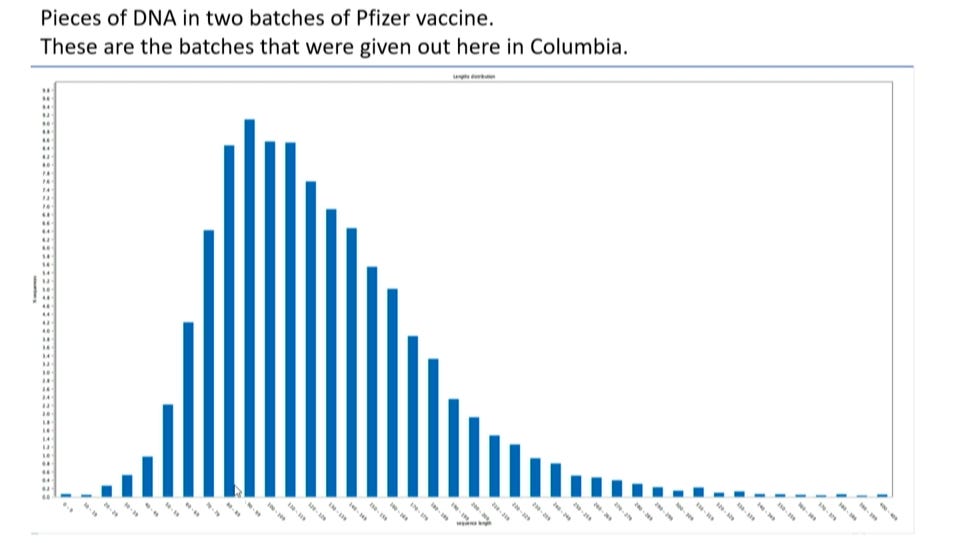
Whereas: As reported earlier in the literature, and presented to the PPLS meeting on September 12, 2023, the mRNA Covid-19 pharmaceuticals were contaminated with DNA segments from the eColi
Whereas: The mechanisms and actions were described by me in the article on substack, establishing how the DNA segments would affect human cells.
An important confirming summary was provided by Dr. Robert Malone
Whereas: The manufacturing and logistics processes for the mRNA Covid-19 pharmaceuticals require stringent logistics controls from the suppliers to vials leaving the manufacturing facility to the shot arriving in the patient’s arm; failure of those controls results in fragments of mRNA and fragments of the LNP to be formed, with unmeasured consequences, but certainly contamination of the original product. See Hedley Rees’s substack (eg, (Rees, 2023))
as well as his book on the requirements for good manufacturing practice (Rees, Taming the Big Pharma Monster, 2022)and pharmaceutical logistics management book (Rees, The Covid-19 Supply Chain: Fact not Fiction). A white paper has been provided to the South Carolina Legislature with more details.
Whereas: The mechanisms, actions, and history of DNA contamination as well as other contamination of mRNA and related vaccines is documented by Dr. Robert Malone in his article (Malone, 2023) The mechanisms of contamination should have been known to the developers, the manufacturers, the state federal and state regulators and advisory organizations, and physicians. This establishes a continuous trail of negligence and malfeasance to all concerned. We recognize that, in some cases, financial penalties would be faced by those who did not cooperate with the program, but this has revealed a level of corruption on immorality that will have to be managed by checks and balances at the state or local level in many future pharma products.
Whereas: The FDA has failed to test and limit, and even facilitated the distribution of, contaminated pharmaceuticals, with the mRNA Covid-19 pharmaceutics being the most obvious.
Therefore, Be It Resolved: A Bill be submitted to the state legislature that establishes a pharmaceutical product quality advisory committee to define and oversee the following process. Sampling of boxes will be required of pharmaceutical products identified by the advisory committee. Sampling of the mRNA Covid-19 pharmaceuticals should be mandatory until it is entirely withdrawn and unavailable in any form in South Carolina. Sampling should be done at the point of administration. Testing should be for any contaminants that might be expected for the manufacturing process of that pharmaceutical. Both effectiveness and adverse effects associated with the pharmaceutical should be considered in identifying other tests that should be run on the sample. If a sample is found contaminated, then a hold should be placed on all lots associated with that box. Other boxes at that location should be more extensively sampled as well as boxes at other locations containing that lot. This quality process should be iterated according to a Poisson sampling algorithm for quality control. If more than one vial or other relevant package of the pharmaceutical is found to not pass quality standards, the entire lot should be recalled. If more than one vial of any other lot was found to not pass quality standards, then the receiving location should be quarantined until a cause is determined. The lots to which the other vial belongs should also be recalled. Recalled lots should be carefully researched to determine the cause of the quality problem (eg, manufacturing, logistics, human handling, etc). Appropriate fines should be established to cover the cost of testing and recovery of potentially defective pharmaceuticals. All information should be recorded in a database that can be merged with EMR data collected at the state level so that adverse effects of the contamination can be tracked. Persons with adverse effects linked to contaminated packages can sue the manufacturer for mislabeled data and collect physical and mental anguish damages in state courts.
Be it Further Resolved A Bill will be submitted to the state legislature creating a pharmaceutical manufacturing quality advisory committee, whose charge should be to create evaluation processes to assess the manufacturing quality of pharmaceutical companies supplying pharmaceuticals to the citizens of the state. Auditing of these companies should be done on a prioritized basis. The audit should consider the manufacturing processes of individual pharmaceuticals as well as good manufacturing practices and company culture as a whole. Those companies that fail the audit could not distribute pharmaceuticals in SC, either through stores or on line for a period of (suggested) one year. After that time, the company can reapply for a quality audit.
Be It Further Resolved that the state should fund both committees and the quality control processes. Committees formed and individuals and organizations performing the testing should have no relationship to pharmaceutical companies, either through grants or past employment. They should not be in any way related to the state’s public health organization. Where possible, the testing should be integrated into community college and university curricula to provide training for students as well as to reduce the cost of sampling and testing..