Bill H4684 Filed in SC House to Permit and Defend Off-Label Use of Pharmaceuticals, Part 2- Bill Discussion History re Ivermectin
The SC Health Department Director Correspondence and Response
Dr. Simmer, Director of the Department of Health and Environmental Control, South Carolina, Sept 12, 2023
On September 12, 2023 the South Carolina legislature held an all-day ad-hoc joint hearing on the performance of the state with respect to Covid-19 era management. The official name of the session was the “Pandemic Preparedness Listening Session.”
The video can be found here:
A summary can be found here:
The biggest news from the meeting resulted in the SV40 and plasmid contamination contraversy, discussed here:
In late October I sent an email to Dr. Simmer that said, in part:
During the September 12 meeting the topic of Ivermectin was discussed by Dr. Simmer, by several committee members, and by some of the speakers. Dr. Simmer, you said:
"My reading of that research is that it (Ivermectin) does not work well for the treatment of Covid. There are multiple studies... those studies have really very consistently not shown that it works for Covid. I wish it did."
This despite what appeared to be the concensus of both committee members and speakers that it did.
Dr. Simmer, you said you would provide the studies you were using to justify your statement. Please send me a list of those studies.
I also said this:
I recommend all go to the website https://c19ivm.org/meta.html. This is a one stop information depot for all studies, good and otherwise, on Ivermectin. Additionally, there are valuable tutorials at the site. Dr. Simmer, what do you think of this web site?
I received a reply last week in the form of a list of studies. The list is:
Popp, M., et. al. Ivermectin for preventing and treating COVID-19. Cochrane Database of Systemic Reviews, 2022 (6): CD015017.
Bramante, C., et. al. Randomized Trial of Metformin, Ivermectin, and Fluvoxamine for Covid-19. New England Journal of Medicine, 2022; 387: 599-610.
Lim, S., et. al. Efficacy of Ivermectin Treatment on Disease Progression Among Adults With Mild to Moderate COVID-19 and Comorbidities: The I-TECH Randomized Clinical Trial. JAMA Internal Medicine, 2022 Apr 1; 182(4): 426-435.
Naggle, S. et. al. Effect of Higher-Dose Ivermectin for 6 Days vs Placebo on Time to Sustained Recovery in Outpatients with COVID-19: A Randomized Clinical Trial. Journal of the American Medical Association, 2023 Mar 21; 329(11): 888-897.
Naggle, S., et. al. Effect of Ivermectin vs Placebo on Time to Sustained Recovery in Outpatients With Mild to Moderate COVID-19. Journal of the American Medical Association, 2022 Oct 25; 328(16); 1595-1603.
Reis, G., et. al. Effect of Early Treatment with Ivermectin among Patients with Covid-19. New England Journal of Medicine, 2022 May 5; 386(18): 1721-1731.
Rezai, M., et. al. Non-effectiveness of Ivermectin on Inpatients and Outpatients with COVID-19; Results of Two Randomized Doble-Blinded, Placebo-Controlled Trials. Frontiers in Medicine, 2022 Jun; 16:9:919708.
If you perform a web search of these using Google or even DuckDuckGo you will get pages of references to these papers and reviews: all positive. But if you go to https://c19ivm.org/meta.html you will get this kind of review for five of the seven:
This was on NaggleNaggie, S., et. al. Effect of Ivermectin vs Placebo on Time to Sustained Recovery in Outpatients With Mild to Moderate COVID-19. Journal of the American Medical Association, 2022 Oct 25; 328(16); 1595-1603.
(In the following, there is a table that didn’t copy over well. It had column headings of “Severity” and “Issue (most recent update 26 days ago”. The column headings are in bold below. The column entry looks like:
CRITICAL
1. Impossible data (31 days ago)
CRITICAL is the severity, and the 1. Impossible data is the Issue.
There is a direct link to this particular review: https://c19ivm.org/activ6ivm.html
Extreme COI, data inconsistencies, uncorrected errors, no response from authors, participant fraud, refusal to release data
SEE ALSO
ACTIV-6 Trial on Ivermectin: NIH Scientists Behaving Badly
The Story Of A Real ACTIV-6 Patient
ACTIV-6 Dosing & Timing: A Fox In The Henhouse
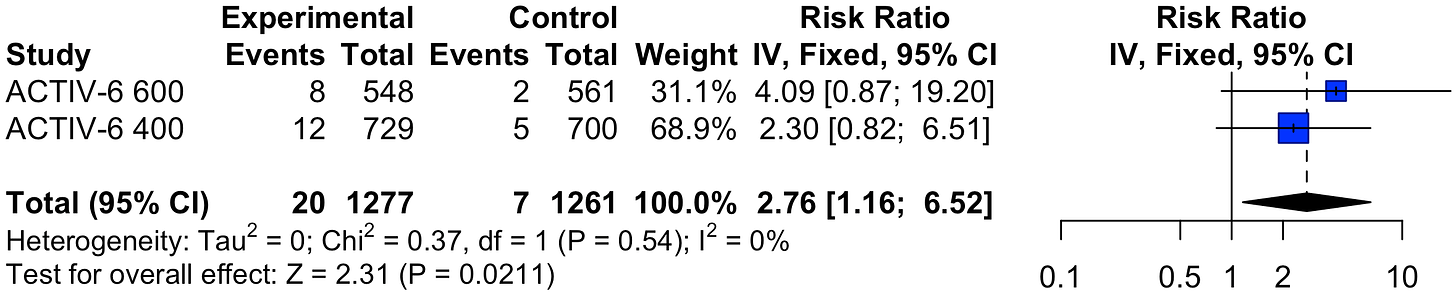
RCT low-risk outpatients with very late treatment (median 6 days, 25% ≥8 days) in the USA, showing 98% probability of efficacy for clinical progression at day 14, a treatment delay-response relationship, and significant efficacy for patients with severe symptoms at baseline. The posterior probability ivermectin is effective was 99%, 98%, 97% for mean time unwell and clinical progression @14 and 7 days. All exceed the pre-specified threshold for superiority fnih.org. Note that the clinical progression results exceeding the superiority threshold in the preprint medrxiv.org changed in the journal version for the 400µg/kg arm, with no explanation for over 410 days). The 600µg/kg arm was reported separately Naggie. When not specified, comments refer to the 400µg/kg arm. We provide more detailed analysis of this study due to widespread incorrect press.
There was one death reported in each of the 400µg/kg and 600µg/kg ivermectin arms. For 400µg/kg, the patient did not take ivermectin. For 600µg/kg, authors note that the death was accidental.
There are many critical issues as below. Design, presentation, and analysis shows a strong negative bias.
Severity
Issue (most recent update 26 days ago)
CRITICAL
1. Impossible data (31 days ago)
CRITICAL
2. Adverse event count mismatch (31 days ago)
CRITICAL
3. Randomization failure - higher severity in ivermectin arms
CRITICAL
4. Adverse events consistent with potential medication error
CRITICAL
5. Adverse events similar for active and placebo
CRITICAL
6. Participant fraud (37 days ago)
CRITICAL
7. Superiority found, not reported
CRITICAL
8. Interim analyses not reported, likely showed superiority (26 days ago)
CRITICAL
9. Death reported in mITT, however participant not in mITT, did not receive study drug
CRITICAL
10. Ivermectin source unknown, specified for other trial medications
CRITICAL
CRITICAL
12. Post-hoc protocol (78 days ago)
CRITICAL
13. Clinical progression results changed
CRITICAL
14. Primary outcome not reported, closest reported outcome shows superiority
CRITICAL
15. Pre-specified primary 14 day outcomes not reported, clinical status shows 30% benefit
CRITICAL
16. Hospitalization/death mismatch
CRITICAL
17. 90 day followup results not provided
CRITICAL
CRITICAL
CRITICAL
20. Patients with symptoms >7 days included
CRITICAL
21. Data unavailable over 540 days from publication
CRITICAL
22. Outcomes reported do not match protocol
CRITICAL
23. Primary outcomes changed after publication
CRITICAL
24. Different hospitalization/urgent care numbers between paper and presentation
CRITICAL
25. Post-hoc primary outcome measured on day 3
CRITICAL
CRITICAL
27. Effective dose ~130μg/kg, administration on empty stomach
CRITICAL
28. Clinical progression details for fluticasone/fluvoxamine but not ivermectin
CRITICAL
29. COVID-19 mortality/hospitalization not reported
CRITICAL
30. Many pre-specified outcomes missing
CRITICAL
31. Full protocol unavailable before October 2022
CRITICAL
32. IDMC not independent, extreme conflict of interest
CRITICAL
33. Reported primary outcome low relevance
CRITICAL
34. Shipping and PCR delays largely enforce late treatment
CRITICAL
CRITICAL
36. Blinding failure
CRITICAL
37. Extreme conflicts of interest
CRITICAL
38. Treatment delay-response relationship
CRITICAL
39. Asymptomatic patients included
CRITICAL
CRITICAL
41. Significant missing data, not mentioned in paper
CRITICAL
42. Statistically significant efficacy for severe patients removed
CRITICAL
43. Statistical analysis plan dated after trial end
CRITICAL
44. 31% more severe cases in the ivermectin arm
CRITICAL
CRITICAL
CRITICAL
47. Author claims results from 596 researchers should be censored for false information
SERIOUS
48. Bias due to false positive antigen tests
SERIOUS
49. Participant pickup delay (37 days ago)
SERIOUS
SERIOUS
SERIOUS
52. No adherence data or per-protocol analysis
SERIOUS
53. Skeptical prior not justified
SERIOUS
54. Not enough tablets provided
SERIOUS
55. Monotherapy with no SOC for most patients
SERIOUS
56. Over 2x greater severe dyspnea at baseline for ivermectin
SERIOUS
57. Safety conclusion removed, suggests bias
SERIOUS
SERIOUS
SERIOUS
60. No breakdown of severe outcomes
SERIOUS
MAJOR
62. Overlapping fluticasone placebo shows very different event numbers
MAJOR
63. Overlapping fluticasone placebo shows unexpected baseline numbers
MAJOR
64. Inconsistent calendar time subgroups
UNKNOWN
65. Outcome graph presented does not match either medication tested
Responses: authors have not responded to any of these issues.
Impossible data. There are major data mismatches twitter.com. For the ivermectin 600 arm clinicaltrials.gov (CT) indicates that 718 patients started and 708 completed the arm clinicaltrials.gov, while the paper claims that only 668 were randomized to ivermectin, of which 66 did not even receive the medication jamanetwork.com. Baseline age, ethnicity, and race data all differ between CT and the paper, for example CT indicates 3 patients in the ivermectin arm were American Indian or Alaska Native, whereas the paper shows 9, despite reporting on a much smaller number of patients. Many results also differ between CT and the paper.
Adverse event count mismatch. The paper reports 44 of 604 placebo patients had an adverse event in the 600µg/kg arm jamanetwork.com, whereas clinicaltrials shows 5 of 724 placebo patients had an adverse event clinicaltrials.gov.
Randomization failure - higher severity in ivermectin arms. The most severe baseline symptom reported is severe dyspnea. For both 600μg/kg and 400μg/kg, the ivermectin arm has higher incidence of severe dyspnea, which is statistically significant across both arms (p = 0.02). This suggests that known and potentially unknown blinding failures are material.
Adverse events consistent with potential medication error. Placebo adverse events are expected to be similar for the 400μg/kg and 600μg/kg arms. The populations are similar and patients are not taking any study treatments. The 600μg/kg arm has a lower overall hospitalization rate. However, the 600μg/kg placebo arm reports 44/604 (7.3%) adverse events, while the 400μg/kg arm reports only 27/774 (3.5%), over 2 times higher. This is a significant increase in adverse events, p = 0.002, without explanation or discussion. Comparing 400μg/kg and 600μg/kg, adverse events are over 2x higher for both the active and placebo arms. The overall increase in adverse events with the higher dosage (total 3x higher) matches expectations, however we expect the increase in the active treatment arm, not both active and placebo arms. One hypothesis is that patient arm classifications are incorrect, i.e., many patients received the opposite of their designated arm. This kind of error is possible in all placebo controlled trials and happened for example in López-Medina (discovered and excluded in that case). This hypothesis is consistent with both the adverse events and the 600μg/kg results, with the very small remaining effect explainable by the 10% non-matching placebo patients. We recommend that participants retain any leftover tablets for analysis.
Adverse events similar for active and placebo. For the 600μg/kg arm authors report that 9% of patients experienced an adverse event, similar to placebo (7%, no significant difference). However significantly more side effects are expected at this dosage (the total dosage is 3x greater due to the longer duration). The 600μg/kg arm in Buonfrate reports much higher adverse events. Overall reporting is higher in this trial, which may include lower severity items, especially within "general disorders". However looking specifically at eye disorders, a known side effect of higher doses of ivermectin, Buonfrate show 46% vs. 3% for ivermectin vs. control. The lack of higher side effects for ivermectin in ACTIV-6 suggests that patients may not have taken authentic ivermectin at the dosage reported. GMK c19ivm.org notes that data was self-reported by patients in ACTIV-6. Highly inaccurate reporting by patients would also apply to the symptomatic results, similarly invalidating the trial.
Participant fraud. A paper on the operation of the trial Lindsell reveals that there was participant fraud - authors identified participants that signed up repeatedly, and participants that withdrew when not randomized to their preferred arm. Authors indicate that they tried to prevent repeat signups but provide no details on the algorithms or the evaluation thereof. It is possible that they only caught a small fraction of the fraud, and possible that improvements to detection were added only later in the trial during or after the ivermectin arms. It is likely that individuals were gaming the system related to the politicization and extreme financial implications. This information was not disclosed previously. Patients were allowed to specify treatments that they accept or decline to be a part of. One author indicates this was most commonly used by patients to specify only ivermectin twitter.com (B), which may be related to fraud targeting ivermectin. There are known paid groups of individuals targeting ivermectin, and it would be simple to bias results towards null without having to break the blinding. Participant fraud has also been reported for a similar remote trial academic.oup.com where known fake surveys were submitted. The self-reported design and absence of professional medical examination opens these kind of trials to participant fraud, which may be significant due to extreme politicization in the study country.
Superiority found, not reported. Day 7 and day 14 clinical progression results and mean time unwell show superiority of ivermectin (note: 400μg/kg arm, preprint version medrxiv.org, clinical progression results were changed without explanation in the journal version). The protocol indicates superiority for OR < 0.9 and posterior probability > 0.95 fnih.org. In the presentation rethinkingclinicaltrials.org, author shows a slide containing these results while stating "this was, um, not statistically significant" (@22:36). These results were seen despite 107 patients having no symptoms at baseline and the use of the skeptical prior doyourownresearch.substack.com
The other Naggie article referenced by Dr. Simmer:
Naggle, S. et. al. Effect of Higher-Dose Ivermectin for 6 Days vs Placebo on Time to Sustained Recovery in Outpatients with COVID-19: A Randomized Clinical Trial. Journal of the American Medical Association, 2023 Mar 21; 329(11): 888-897.
was reviewed on the website by pointing to a couple of substack posts on doyourownresearch.substack.com. In order to package up a response to Dr. Simmer’s response that I sent to the PPLS committee, I created a PDF file of the two relevant posts:
The articles show the relationship of the Activ-6 protocols to the drug companies and NIH (specifically Fauci) and the poor design of the protocols.
The conclusion you can see for yourself is that all seven papers Dr. Simmer provided had disqualifying flaws in the protocols and execution of those protocols. Dr. Simmer, the Director of South Carolina’s Department of Health and Environmental Control failed to reference one properly designed and executed reference.
I packaged the above documentation into PDFs and I said, in part, in my cover letter to the PPLS committee:
(The following is referencing the video of the September 12 PPLS meeting:)
In one video segment the exchange is:
Corbin: "Somebody as learned as yourself..."
Simmer: "Yep"
Corbin: "The studies, and you referenced several studies on Ivermectin and HCQ that were inconclusive... don't you think that they might be presented that way in order to get a new drug that is much more profitable out among the populus like, say, the vaccine?"
Simmer: "If those studies were done by the pharmaceutical company, I would be very worried about bias. Yes. But they weren't. Moderna and Pfizer were not doing the Ivermectin studies. Those were done independently... and I can assure you... one of the things that I did as part of my Masters in Public Health is learning how to take a study apart and decide if it was reliable or not.”
Corbin: Well, who funded the studies? Did you look at that? They might not have been done by the pharmaceutical companies, but were they funded in any way by the pharmaceutical companies?
Simmer: “No. The studies that I have seen were not funded by the pharmaceutical companies. They had a variety of funding sources, some public, some private…”
Corbin: “Because I looked at some studies of Ivermectin that (inaudible) different conclusion that what you had, that showed some decent effectiveness ..(inaudible) all these different studies, and you’re balancing plates, and we’re just trying to get to the truth and at the end of the day everybody up here and I’m sure everybody in this room loves South Carolina and the people in it… we just want to do what is best and get to the truth…”
In another video segment of the September 12 PPLS meeting, Sen Garrett had this exchange:
Garrett: “Your opinion on HCQ, on Ivermectin… is it an effective treatment for Covid?…”
Simmer: I can tell you that there are many studies…the last time I testified in a hearing like this my comment was that for Ivermectin we needed more research. A lot of that research has now been done… As much as I wish it had turned out otherwise, my reading of that research is that it does not work well for Covid, for treatment of Covid.
Garrett: “It does not work well…”
Simmer: It does not work well for the treatment of Covid. There are a num… there are multiple studies that show that. And they looked at various things: patients in the hospital, patients not in the hospital, patients with mild disease, severe disease. So there are a number ways that they’ve gone at this. But those studies have really very consistently not shown that it works for Covid. Unfortunately. I wish it did. … What I would say for Ivemectin (inaudible) it can be safe, in the human dosage. I certainly discourage people from taking the animal dosage.”
My email continues:
The Committee can see that all seven studies were between fraudulent and extremely unreliable. The studies were all funded/supported and the protocols influenced by the Federal government. See the influences yourself in the summary. You will see that Dr. Simmer's response was a misdirection. Examine the Federal Government's influence on Dr. Simmer's recommended research articles. I can provide details of both the money and the "authority" flow if needed. Pharma's fingerprints are all over every step in the research, with the Federal Government laundering the money. On the other hand, I personally don't hold the pharmas as responsible for the Covid-19 disaster as much as I hold the Federal Government responsible. They protect the pharmas and create the legal foundation for the misdeeds. The South Carolina legislature must protect South Carolinians from the Federal Government.
By his own testimony, Dr. Simmer appears to be confident and proud of his credentials and his expertise. That he provided some of the worst experimental process examples available is remarkable. If we believe Dr. Simmer, we have to suspect that his motivation in selecting them must have been nefarious. Somebody or something must have dictated that he go against his proclaimed expertise. Was it contracts? Was it blackmail? Was it CrowdThink?
After a couple of paragraphs regarding legal actions that might be taken, I end with:
I believe it is essential for the PPLS committee to understand the motive behind Dr. Simmer's testimony and latest response. In management terminology, the PPLS members must discover the root cause. This is absolutely essential, because it is clear the legislature is going to be responsible for defining the protections to prevent this ever happening again.
We will see what the PPLS committee does with this information. In the meantime, any support from experts for Bill H4684 from experts anywhere in the world would be appreciated. Comments below. (Likes won’t do.)